Research Insight
Public Perception and Acceptance of Genetically Engineered Pigs in Medical Applications: Strategies for Effective Communication 
 Author
Author  Correspondence author
Correspondence author
GMO Biosafety Research, 2024, Vol. 15, No. 5
Received: 15 Jul., 2024 Accepted: 26 Aug., 2024 Published: 08 Sep., 2025
This study explores public perception and acceptance of genetically modified (GM) pigs in medical applications, systematically analyzing key factors influencing public attitudes, including technological awareness, ethical and cultural values, and risk perception differences. The research finds that the shortage of xenotransplantation donors has driven the rapid development of GM pigs in the medical field, including applications in organ transplantation, biopharmaceutical production, and disease modeling. However, alongside these medical breakthroughs, the technology has sparked ethical controversies and public concerns over safety risks, making public acceptance a critical variable affecting its implementation and adoption. Significant differences in acceptance exist among various groups—medical professionals, the general public, and patients' families. Information sources, trust mechanisms, and ethical sensitivities play important roles in shaping acceptance. To improve public acceptance, this study proposes effective science communication strategies, including establishing participatory communication mechanisms, adopting diversified dissemination formats and narrative strategies, and strengthening institutional transparency and third-party oversight. This research offers practical significance for enhancing public understanding and support for the medical application of GM pigs, and for promoting coordinated progress in medical innovation and ethical governance.
1 Introduction
The global shortage of human organs for transplantation has intensified the search for alternative solutions, with xenotransplantation—particularly the use of genetically modified (GM) pigs—emerging as a promising avenue to address this critical medical need. Advances in genetic engineering have enabled the development of pigs whose organs are more compatible with human physiology, significantly improving the prospects for successful transplantation and reducing immunological barriers. However, these scientific breakthroughs are accompanied by complex ethical debates and societal concerns, making public perception and acceptance pivotal to the future implementation of this technology (Cooper et al., 2016; Cooper et al., 2018; Hryhorowicz et al., 2020; Xi et al., 2023).
Public attitudes toward the use of GM pigs in medical applications are shaped by a range of factors, including perceived benefits, potential risks, ethical considerations, and the level of understanding of genetic technologies (Yunes et al., 2019). As such, effective communication strategies are essential to foster informed public dialogue, address misconceptions, and build trust in the safety and necessity of xenotransplantation.
This study will summarize existing research on public perception and acceptance of GM pigs for medical purposes and propose evidence-based communication pathways to increase public understanding and acceptance, thereby supporting the responsible incorporation of GM pig-derived medical solutions into clinical practice. This study aims to combine scientific innovation with societal values to inform policy and practice in the evolving field of xenotransplantation.
2 Background of GM Pig Applications in Medicine
2.1 Main application areas
Genetically modified pigs are the leading candidates for organ and tissue donation in xenotransplantation, addressing the critical shortage of human organs. Organs such as the heart, kidney, islets, and corneas from GM pigs have demonstrated prolonged survival and function in non-human primate models, and recent clinical trials have begun to test these organs in human recipients (Cooper et al., 2018; Montgomery et al., 2022; Wolf et al., 2023; Peterson et al., 2024). Genetic modifications are designed to overcome immunological barriers, reduce the risk of rejection, and minimize the transmission of porcine pathogens (Cooper et al., 2016; Sykes and Sachs, 2019; Lei et al., 2022; Xi et al., 2023; Peterson et al., 2024).
GM pigs are also engineered to serve as bioreactors, producing high-quality recombinant proteins, therapeutic antibodies, and even humanized insulin for medical use. This application leverages the pig’s physiology to generate complex proteins that are difficult to produce in other systems, offering new avenues for treating diseases such as diabetes (Figure 1) (Yang et al., 2016; Hryhorowicz et al., 2020).
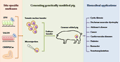 Figure 1 Figure 1 Schematic diagram of generation genetically engineered pig for biomedical purposes (Adopted from Hryhorowicz et al., 2020) Image caption: The different site-specific nucleases (ZFN, TALEN, CRISPR/Cas9) used for genome editing and two techniques (somatic nuclear transfer and microinjection) to produce genetically modified pigs are shown. Biomedical applications for which genetically engineered pigs are generated include modeling human diseases, production of pharmaceutical proteins, and xenotransplantation (Adopted from Hryhorowicz et al., 2020) |
Due to their anatomical and physiological similarities to humans, genetically engineered pigs are increasingly used as models for human diseases, including cardiovascular diseases, cancers, diabetes, neurodegenerative disorders, and rare genetic conditions. These models enable more accurate preclinical testing and the development of new therapies and vaccines (Prather et al., 2013; Wolf et al., 2014; Holm et al., 2016; Perleberg et al., 2018; Kalla et al., 2020).
2.2 Current research progress and clinical trials
The advent of precision gene-editing tools, particularly CRISPR/Cas9, has revolutionized the creation of GM pigs. These technologies allow for targeted modifications that enhance immunocompatibility, reduce the risk of organ rejection, and improve the safety profile of pig-derived medical products. Multiple gene edits can now be introduced efficiently, enabling the production of pigs with complex genetic modifications tailored for specific medical applications (Ryu et al., 2018; Sykes and Sachs, 2019; Tanihara et al., 2021; Peterson et al., 2024).
Recent clinical studies have reported successful transplantation of kidneys from genetically modified pigs into brain-dead human recipients, with the organs functioning well and showing no signs of hyperacute rejection over several days. These results mark a significant milestone toward the clinical adoption of xenotransplantation and highlight the potential of GM pigs to address the organ shortage crisis (Lei et al., 2022; Montgomery et al., 2022; Wolf et al., 2023; Peterson et al., 2024).
3 Public Perception and Acceptance Analysis
3.1 Core factors influencing public acceptance
Public acceptance is not strongly linked to technical knowledge of genetic modification; even those with limited understanding may form opinions based on perceived benefits or risks rather than scientific details. Misconceptions and low awareness are common, suggesting that knowledge alone does not drive acceptance (Yunes et al., 2019; Andrés-Sánchez et al., 2025).
Attitudes are shaped by ethical concerns about species boundaries, animal rights, and perceptions of what is “natural.” Many people express discomfort with genetic modification due to beliefs about tampering with nature or the moral status of animals. Cultural context and personal values play a significant role, with some groups more likely to oppose GM pigs on ethical grounds (Jin et al., 2022; Van Eenennaam, 2024).
Concerns about biosafety, cross-species virus transmission, and unforeseen long-term consequences are major barriers to acceptance. Risk perception is a strong negative predictor of support, often outweighing technical understanding (Jin et al., 2022; Van Eenennaam, 2024). Requests for clarification of risks are common among those hesitant to support GM pig applications (Yunes et al., 2019).
3.2 Acceptance differences among groups
Medical professionals and those directly affected by organ shortages (e.g., patient families) may be more accepting of GM pig applications due to perceived medical benefits, while the general public tends to be more cautious or skeptical (Crane et al., 2020; Van Eenennaam, 2024).
Acceptance does not consistently vary by age, religion, or education, though some studies note that those from agricultural backgrounds may be less accepting (Yunes et al., 2019). General attitudes and affective responses-rather than demographic factors-are stronger predictors of acceptance (Jin et al., 2022; Andrés-Sánchez et al., 2025).
3.3 Sources of information and trust mechanisms
Public opinion is shaped by a mix of traditional media, social networks, and scientific institutions. Misinformation and affective responses in media can amplify concerns or biases, while transparent communication from trusted scientific sources can improve acceptance (Jin et al., 2022; Van Eenennaam, 2024).
Cognitive biases, such as generalizing negative attitudes toward all GM applications, and a lack of trust in institutions or regulatory bodies, can hinder acceptance (Jin et al., 2022; Van Eenennaam, 2024; Andrés-Sánchez et al., 2025). Addressing these biases and building trust through transparent, evidence-based communication is essential for improving public attitudes.
4 Major Challenges in Science Communication and Public Engagement
4.1 Information complexity and public understanding gap
The technical complexity of genetic engineering and xenotransplantation creates a significant barrier for lay audiences. High-tech jargon and specialized knowledge are difficult to translate into accessible language, often resulting in confusion or misunderstanding among the public. This “expert dominance” in medical and scientific discourse can further widen the communication gap, as information is frequently tailored to professional audiences rather than the general public. As a result, the broader societal implications and potential benefits or risks of genetically engineered pigs may not be fully understood or appreciated by non-experts, highlighting the need for social science perspectives and inclusive communication strategies (Kögel et al., 2024a).
4.2 Managing sensitivity of ethical debates
Ethical concerns are central to public debates about genetically engineered pigs, particularly regarding the creation and use of animals for human benefit. These debates often involve deeply held beliefs about species boundaries, animal welfare, and the sanctity of life, which can be influenced by religious and cultural values. The unpredictability of social and personal consequences for patients and their families adds further complexity to these discussions. Engaging with these ethical dilemmas requires sensitivity and respect for diverse viewpoints, as well as transparent dialogue that acknowledges both the potential benefits and moral challenges of biotechnology (Kögel et al., 2024a).
4.3 Spread of panic and misinformation risks
The proliferation of fragmented and emotional narratives on social media can amplify public fears and spread misinformation about genetically engineered pigs and their medical applications. Sensationalized stories and conspiracy theories may disrupt informed public discourse, erode trust in science and policy, and hinder the development of evidence-based regulations. This environment makes it challenging for accurate, balanced information to reach the public, emphasizing the importance of proactive, transparent communication and community engagement to counteract misinformation and build trust (Neuhaus, 2018; Kögel et al., 2024a).
5 Effective Communication Strategies and Practical Pathways
5.1 Establishing a public-centered dialogue mechanism
Moving from one-way dissemination to two-way, participatory communication is essential. Engaging patients, families, religious groups, and other stakeholders in open discussions about the science, ethics, and societal implications of genetically engineered pigs can help bridge understanding gaps and foster trust. Such dialogue mechanisms ensure that diverse perspectives are heard and integrated into policy and practice, rather than allowing expert voices to dominate and potentially alienate the public (Xi et al., 2023).
5.2 Diversified communication formats and narrative design
To make complex scientific concepts accessible, communication should employ analogies and storytelling that resonate emotionally and intellectually with lay audiences. Visual media—including infographics, short videos, and interactive exhibitions—can further enhance engagement and comprehension, especially for those with limited technical backgrounds. These approaches help demystify genetic engineering and highlight its real-world benefits, making the science more relatable and memorable (Perleberg et al., 2018; Xi et al., 2023).
5.3 Building trust-based institutional communication systems
Transparency from research institutions, government agencies, and medical organizations is fundamental to building public trust. Clear communication about research goals, safety protocols, and regulatory oversight can address fears and skepticism. Introducing third-party oversight, such as independent ethics committees or public advisory boards, can further increase credibility and openness, reassuring the public that ethical and safety standards are rigorously maintained (Xi et al., 2023).
6 Case Studies in Public Communication and Science Engagement
6.1 Public acceptance cases in xenotransplantation
Successful public engagement in xenotransplantation has involved transparent dissemination of clinical trial information and proactive inclusion of diverse community perspectives. For example, the United States has seen public dissemination of experimental pig kidney and heart xenotransplantation cases, which has underscored the importance of ongoing public engagement and the need to address concerns meaningfully. These efforts have highlighted the value of empirical studies on public opinion, including attention to racial, ethnic, and religious differences, and have recommended that biotechnology companies take a leading role in funding public engagement research (Hurst and Cooper, 2024). In Canada, a nation-wide public consultation on islet xenotransplantation was conducted, representing an “experimental democracy” approach that allowed for broad public input and temporarily increased trust and transparency (Cheng, 2015).
Conversely, failures in public communication have often stemmed from insufficient engagement or lack of transparency. In Australia, public input was sought through a national inquiry, but both Canada and Australia ultimately imposed temporary moratoriums on clinical xenotransplantation due to unresolved public concerns and regulatory uncertainty. These cases illustrate that when public fears—especially regarding disease transmission and ethical issues—are not adequately addressed, policy responses may default to caution, stalling scientific progress (Cheng, 2015). Additionally, a lack of attention to minority religious viewpoints and underrepresented communities can exacerbate mistrust and hinder acceptance (Gusmano, 2022; Hurst and Cooper, 2024).
6.2 Public perception of GM pharmaceutical production
While direct case studies on GM pharmaceutical production are less detailed in the current literature, parallels can be drawn from xenotransplantation. Public acceptance is closely tied to transparent communication, ethical framing, and the inclusion of diverse perspectives in decision-making processes. Social science research emphasizes the need to address both the technical and ethical dimensions of GM biopharmaceuticals to foster acceptance (Kögel et al., 2024a; Kögel et al., 2024b).
Key lessons include the necessity of clear, accessible information, the importance of engaging with ethical and cultural concerns, and the value of participatory dialogue. Regulatory transparency and third-party oversight are also critical for building public trust. These strategies, proven effective in xenotransplantation, are equally applicable to GM pharmaceutical production, where public skepticism can be mitigated through inclusive and transparent engagement (Cheng, 2015; Hurst and Cooper, 2024; Kögel et al., 2024a).
7 Coordination at Policy and Education Levels
7.1 Strategies for enhancing public scientific literacy
Introducing bioethics and gene technology content into school curricula and public science education is a foundational step toward improving scientific literacy. Early and accessible education can demystify genetic engineering, address misconceptions, and prepare future generations for informed participation in societal debates about biotechnology. Public awareness and acceptance are recognized as key factors in the successful transition of genetically modified products from the laboratory to real-world applications, underscoring the importance of long-term, multidomain educational efforts (Chen et al., 2025).
Establishing public participation platforms for technology assessment (pTA) further empowers citizens to engage in the evaluation and oversight of emerging biotechnologies. These platforms can facilitate dialogue between scientists, policymakers, and the public, ensuring that diverse perspectives are considered in decision-making processes and that societal values are reflected in policy outcomes (Chen et al., 2025).
7.2 Ethical regulation for technological advancement
Defining clear boundaries for the medical use of pig-derived materials and establishing robust frameworks for the welfare of genetically modified animals are critical for ethical progress. Policies and regulations must ensure that the use of genetically engineered pigs in medicine is both scientifically justified and ethically acceptable. This includes developing guidelines that address animal welfare concerns, such as minimizing pain and suffering, and considering options like genetic disenhancement to further reduce animal distress when alternatives are unavailable (Rodger et al., 2024).
Regulatory agencies play a pivotal role in developing and enforcing these ethical and legal frameworks. Ongoing policy development, informed by long-term research and public input, is necessary to create transparent, credible, and ethically sound pathways for the application of genetically engineered pigs in medicine (Rodger et al., 2024; Chen et al., 2025).
8 Concluding Remarks
Genetically engineered pigs offer significant promise for addressing critical shortages in organ transplantation and advancing biomedical research. Their physiological similarities to humans and the ability to tailor genetic modifications have positioned pigs as valuable models for human disease and as potential sources for organs, tissues, and pharmaceuticals. However, despite these scientific advances, public perception and ethical acceptance remain substantial challenges.
Establishing diverse trust mechanisms is essential for social acceptance. Transparent regulatory oversight, third-party evaluation, and clear communication from research institutions and government agencies can help build public confidence in the safety and ethical management of genetically engineered pigs for medical use . Effective scientific storytelling-using accessible language, relatable analogies, and emotionally resonant narratives-can bridge the gap between complex science and public understanding, making the benefits and risks more tangible to non-experts.
Participatory communication channels, which actively involve patients, families, religious groups, and other stakeholders in dialogue and decision-making, are key to addressing ethical concerns and fostering a sense of shared responsibility. These approaches ensure that diverse perspectives are considered and that public values are integrated into policy and practice.
Ultimately, the integration of science communication, public engagement, and coordinated policy development forms the foundation for the responsible and socially accepted advancement of genetically engineered pigs in medicine. Continued efforts in these areas will be critical for realizing the full potential of this technology while maintaining public trust and ethical integrity.
Acknowledgments
Thank you to the anonymous peer review for providing targeted revision suggestions for the manuscript.
Conflict of Interest Disclosure
The author affirms that this research was conducted without any commercial or financial relationships that could be construed as a potential conflict of interest.
Andrés-Sánchez J.D., Puelles-Gallo M., Souto-Romero M., and Arias-Oliva M., 2025, Importance–performance map analysis of the drivers for the acceptance of genetically modified food with a theory of planned behavior groundwork, Foods, 14(6): 932.
https://doi.org/10.3390/foods14060932
Chen J., Wang J., Zhao H., Tan X., Yan S., Zhang H., Wang T., and Tang X., 2025, Molecular breeding of pigs in the genome editing era, Genetics Selection Evolution, 57(1): 12.
https://doi.org/10.1186/s12711-025-00961-7
Cheng M., 2015, Islet xeno/transplantation and the risk of contagion: local responses from Canada and Australia to an emerging global technoscience, Life Sciences, Society and Policy, 11(1): 12.
https://doi.org/10.1186/s40504-015-0030-2
Cooper D., Ekser B., Ramsoondar J., Phelps C., and Ayares D., 2016, The role of genetically engineered pigs in xenotransplantation research, The Journal of Pathology, 238(2): 288-299.
https://doi.org/10.1002/path.4635
Cooper D., Gaston R., Eckhoff D., Ladowski J., Yamamoto T., Wang L., Iwase H., Hara H., Tector M., and Tector A., 2018, Xenotransplantation—the current status and prospects, British Medical Bulletin, 125(1): 5-14.
https://doi.org/10.1093/bmb/ldx043
Crane A., Shen F., Brown J., Cormack W., Ruíz-Estévez M., Voth J., Sawai T., Hatta T., Fujita M., and Low W., 2020, The American public is ready to accept human–animal chimera research, Stem Cell Reports, 15: 804-810.
https://doi.org/10.1016/j.stemcr.2020.08.018
Gusmano M., 2022, Xenotransplantation clinical trials and the need for community engagement, The Hastings Center Report, 52(5): 42-43.
https://doi.org/10.1002/hast.1420
Holm I., Alstrup A., and Luo Y., 2016, Genetically modified pig models for neurodegenerative disorders, The Journal of Pathology, 238(2): 267-287.
https://doi.org/10.1002/path.4654
Hryhorowicz M., Lipinski D., Hryhorowicz S., Nowak-Terpiłowska A., Ryczek N., and Zeyland J., 2020, Application of genetically engineered pigs in biomedical research, Genes, 11(6): 670.
https://doi.org/10.3390/genes11060670
Hurst D., and Cooper D., 2024, The importance of public engagement in clinical xenotransplantation, Health Care Science, 3: 124-130.
https://doi.org/10.1002/hcs2.91
Jin S., Li W., Dawson I., Clark B., Chen S., and Frewer L., 2022, Consumer responses to genetically modified food in China: the influence of existing general attitudes, affect and perceptions of risks and benefits, Food Quality and Preference, 99: 104543.
https://doi.org/10.1016/j.foodqual.2022.104543
Kalla D., Kind A., and Schnieke A., 2020, Genetically engineered pigs to study cancer, International Journal of Molecular Sciences, 21(2): 488.
https://doi.org/10.3390/ijms21020488
Kögel J., Cook P., Brown N., Clare A., Glick M., Hansson K., Idvall M., Lundin S., Michael M., Rogvi S., and Sharp L., 2024a, Engineering organs, hopes and hybridity: considerations on the social potentialities of xenotransplantation, Medical Humanities, 51(1): 180-184.
https://doi.org/10.1136/medhum-2024-013061
Kögel J., Ernst P., Sauermeister J., and Marckmann G., 2024b, Ethical implications of social science research on xenotransplantation, Xenotransplantation, 31(6): e70004.
https://doi.org/10.1111/xen.70004
Lei T., Chen L., Wang K., Du S., Gonelle-Gispert C., Wang Y., and Buhler L., 2022, Genetic engineering of pigs for xenotransplantation to overcome immune rejection and physiological incompatibilities: the first clinical steps, Frontiers in Immunology, 13: 1031185.
https://doi.org/10.3389/fimmu.2022.1031185
Montgomery R., Stern J., Lonze B., Tatapudi V., Mangiola M., Wu M., Weldon E., Lawson N., Deterville C., Dieter R., Sullivan B., Boulton G., Parent B., Piper G., Sommer P., Cawthon S., Duggan E., Ayares D., Dandro A., Fazio-Kroll A., Kokkinaki M., Burdorf L., Lorber M., Boeke J., Pass H., Keating B., Griesemer A., Ali N., Mehta S., and Stewart Z., 2022, Results of two cases of pig-to-human kidney xenotransplantation, The New England Journal of Medicine, 386(20): 1889-1898.
https://doi.org/10.1056/NEJMoa2120238
Neuhaus C., 2018, Community engagement and field trials of genetically modified insects and animals, The Hastings Center Report, 48(1): 25-36.
https://doi.org/10.1002/hast.808
Perleberg C., Kind A., and Schnieke A., 2018, Genetically engineered pigs as models for human disease, Disease Models & Mechanisms, 11(1): dmm030783.
https://doi.org/10.1242/dmm.030783
Peterson L., Yacoub M., Ayares D., Yamada K., Eisenson D., Griffith B., Mohiuddin M., Eyestone W., Venter J., Smoleński R., and Rothblatt M., 2024, Physiological basis for xenotransplantation from genetically modified pigs to humans, Physiological Reviews, 104: 1409-1459.
https://doi.org/10.1152/physrev.00041.2023
Prather R., Lorson M., Ross J., Whyte J., and Walters E., 2013, Genetically engineered pig models for human diseases, Annual Review of Animal Biosciences, 1: 203-219.
https://doi.org/10.1146/annurev-animal-031412-103715
Rodger D., Hurst D., Bobier C., and Symons X., 2024, Genetic disenhancement and xenotransplantation: diminishing pigs’ capacity to experience suffering through genetic engineering, Journal of Medical Ethics, 50: 729-733.
https://doi.org/10.1136/jme-2023-109594
Ryu J., Prather R., and Lee K., 2018, Use of gene-editing technology to introduce targeted modifications in pigs, Journal of Animal Science and Biotechnology, 9(1): 5.
https://doi.org/10.1186/s40104-017-0228-7
Sykes M., and Sachs D., 2019, Transplanting organs from pigs to humans, Science Immunology, 4(41): eaau6298.
https://doi.org/10.1126/sciimmunol.aau6298
Tanihara F., Hirata M., and Otoi T., 2021, Current status of the application of gene editing in pigs, The Journal of Reproduction and Development, 67: 177-187.
https://doi.org/10.1262/jrd.2021-025
Van Eenennaam A., 2024, 120 gene editing: from the general public perspective, Journal of Animal Science, 120(2): 14.
https://doi.org/10.1093/jas/skae102.018
Wolf E., Reichart B., Moretti A., and Laugwitz K., 2023, Designer pigs for xenogeneic heart transplantation and beyond, Disease Models & Mechanisms, 16(5): dmm050177.
https://doi.org/10.1242/dmm.050177
Wolf E., Braun-Reichhart C., Streckel E., and Renner S., 2014, Genetically engineered pig models for diabetes research, Transgenic Research, 23: 27-38.
https://doi.org/10.1007/s11248-013-9755-y
Xi J., Zheng W., Chen M., Zou Q., Tang C., and Zhou X., 2023, Genetically engineered pigs for xenotransplantation: hopes and challenges, Frontiers in Cell and Developmental Biology, 10: 1093534.
https://doi.org/10.3389/fcell.2022.1093534
Yang Y., Wang K., Wu H., Jin Q., Ruan D., Zhen O., Zhao B., Liu Z., Zhao Y., Zhang Q., Fan N., Liu Q., Guo S., Bu L., Fan Y., Sun X., Li X., and Lai L., 2016, Genetically humanized pigs exclusively expressing human insulin are generated through custom endonuclease-mediated seamless engineering, Journal of Molecular Cell Biology, 8(2): 174-177.
https://doi.org/10.1093/jmcb/mjw008
Yunes M., Teixeira D., von Keyserlingk M., and Hötzel M., 2019, Is gene editing an acceptable alternative to castration in pigs?, PLOS ONE, 14(6): e0218176.
https://doi.org/10.1371/journal.pone.0218176

. HTML
Associated material
. Readers' comments
Other articles by authors
. Yeping Han
Related articles
. Genetically modified pigs
. Xenotransplantation
. Public perception
. Ethical controversy
. Science communication
Tools
. Post a comment


